GeneDx Holdings (WGS)·Q4 2025 Earnings Summary
GeneDx Beats Q4 on 34% Volume Surge, Guides 30% Revenue Growth for 2026
February 23, 2026 · by Fintool AI Agent
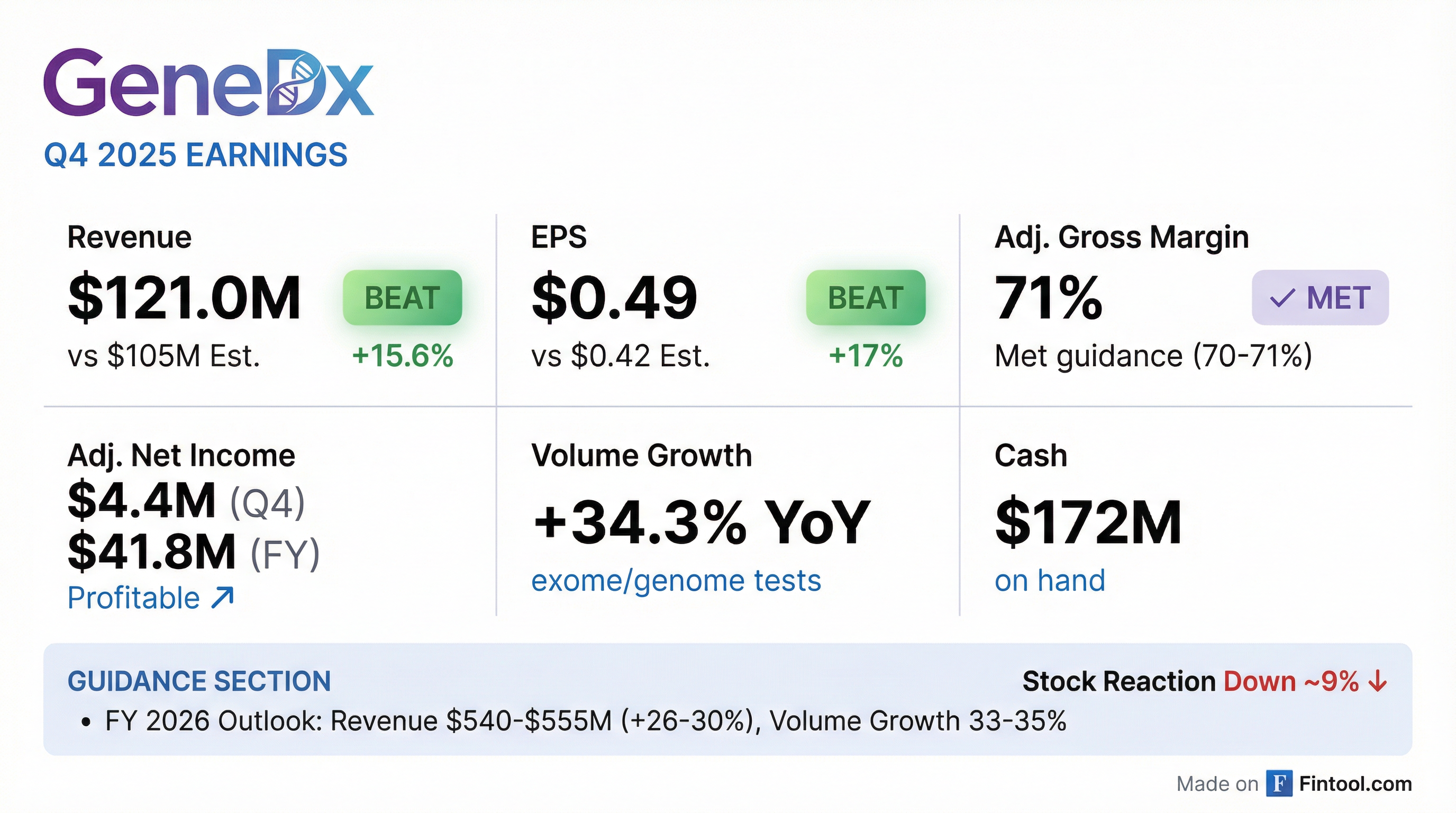
GeneDx (WGS) delivered another strong beat in Q4 2025, with revenue of $121.0 million exceeding consensus by 15.6% and adjusted EPS of $0.49 topping estimates by 17% . The rare disease diagnostics leader reported exome and genome test volume growth of 34.3% year-over-year, reaching 27,761 tests in the quarter . Full-year 2025 revenue came in at $427.5 million, meeting the high end of guidance, with adjusted net income of $41.8 million . Despite the beat, shares fell ~9% as investors digest the company's aggressive investment plans for its general pediatrics expansion.
Did GeneDx Beat Earnings?
GeneDx delivered a convincing double-beat in Q4 2025:
*Values retrieved from S&P Global
The beat was driven by:
- Accelerating volume growth: Q4 volume of 27,761 exome/genome tests, up 34.3% YoY
- Strong reimbursement: Continued improvement in payment rates and denial reductions
- Mix shift: Ongoing conversion from panels to higher-value exome/genome testing
What Did Management Guide for 2026?
GeneDx provided robust FY 2026 guidance that implies continued strong momentum:
Management broke down the expected growth contribution by market layer :
- Foundational Markets (Geneticists, Pediatric Specialists): 25-27%
- Expansion Markets (NICU, Prenatal, Adult Specialists, International): 7-8%
- Future Markets (gNBS, BioPharma, New Channels): 1%

How Did the Stock React?
Despite the strong beat, WGS shares fell approximately 9% following the earnings release. The stock closed at $82.06, down from $90.68, with aftermarket trading showing modest recovery to $83.51.
The negative reaction likely reflects several factors:
- Profit-taking: WGS had run up significantly over the past year
- Investment cycle concerns: Management signaled continued OpEx investment ahead of general pediatrics ramp
- Deceleration optically: 2026 revenue growth guidance of 26-30% is below 2025's 40%+ growth rate, though still strong
- Extended timeline: The 18-24 month adoption timeline for general pediatrics from June 2025 AAP guidelines means meaningful volume contribution won't materialize until late 2026/early 2027
What Changed From Last Quarter?
Key developments since Q3 2025:
Strategic progress:
- FDA Breakthrough Designation: Received for ExomeDx and GenomeDx tests, validating GeneDx Infinity database as gold standard
- Medicaid Expansion: Secured coverage in 8 new states (including CA, CO, OH), bringing total states covering exome/genome to 38 and NICU rapid testing coverage to 17 states
- Commercial Buildout: Hiring dedicated GenPeak sales team to target 25,000 general pediatricians
Key KPIs and Trends
GeneDx has delivered 8 consecutive quarters of beats, with volume growth accelerating:
The company's exome and genome testing now represents 84% of total revenue, up from 67% in 2022 .
Growth Strategy Deep Dive
GeneDx's growth flywheel is built on the GeneDx Infinity database—the largest rare disease dataset with nearly 1 million exomes and genomes and over 7 million phenotypic data points .
Current Market Positioning:
- 80% market share among clinical geneticists, only ~30% patient penetration—room to grow
- ~30% clinician penetration in pediatric neurology, 15% patient penetration after 3 years
- FDA Breakthrough Device designation positioning GeneDx as first FDA-authorized comprehensive genomic solution
- GeneDx Infinity: 2.5M+ rare genetic tests, 1M+ exomes/genomes, 8M+ phenotypic data points
Expansion Market Details:
- General Pediatrics (50 dedicated reps deployed January 2026): "One-minute ordering" workflow launching end of Q2/early Q3, with volume expected to pick up Q4 2026
- NICU (10 reps + new CMO Dr. Linda Genen): ~25% of target accounts are existing customers but utilization in single digits—aiming for 60% over time. Should be ordering 200K+ tests annually
- Prenatal (10 reps, launched Q1): Targeting MFM specialists for patients with abnormal ultrasounds. Expect 0 contribution Q1, little Q2, nice ramp Q3+
- Adult Specialists: Leveraging specialist sales force for adult neurologists diagnosing pediatric-onset conditions missed in childhood
- International (5 reps): Software and Interpretation-as-a-Service model, late 2026+ contributor
Q&A Highlights
On Q1 2026 Expectations: CFO Kevin Feeley set the baseline at 33% volume growth for Q1, implying a sequential decline of 300-400 tests from Q4—standard seasonality given deductible resets and weather impacts . "Q1 always steps down due to deductible reset dynamics... we factored this rhythm into our guidance" .
On Competition: CEO Katherine Stueland was direct about GeneDx's competitive moat: "Our sales team is 6 times the next largest sales team. I think we have more reps in California than one company has for their entire sales team" . She emphasized that Infinity delivers "2 times the accuracy of another exome or genome out on the market today" with turnaround times of ~2 weeks vs. competitors who take "far longer" .
On Reimbursement Conservatism: Management baked in a 70% denial rate assumption for new outpatient call points—mirroring their experience when pediatric neurology launched in 2023. "We've taken that past experience and applied that level of expectation for the first few quarters out of the gate" .
On NICU Momentum: Early Q1 2026 signs are encouraging. Stueland noted they're "seeing positive signs from an ordering perspective year to date" and are "taking a look at do we want to add more reps" . New CMO Dr. Linda Genen (a neonatologist) is driving peer-to-peer KOL strategy.
On Medicaid: Nothing baked into guidance for Medi-Cal (despite November policy effective date) or any new Medicaid state expansion: "The guide does effectively assume zero new Medicaid states this year. Do I think that that's reasonable to expect zero? I don't, but we'll always leave new Medicaid coverage out until proven otherwise" .
Key Management Quotes
"2026 is going to be a breakout year for GeneDx. We're operating in an enormous and largely untapped market with a 25-year head start." — Katherine Stueland, CEO
"Competition is a good thing... it validates [the market], but it also helps put more and different types of pressure on clinicians to start using this testing." — Katherine Stueland
"Infinity was built specifically for rare disease, patient by patient, year by year in the clinic. It would take decades to replicate what we have today, and by then, we'll be decades further ahead." — Katherine Stueland
Pricing and Margin Progression
GeneDx has demonstrated strong pricing power with average reimbursement rate (ARR) and gross margin expanding significantly:
CFO Kevin Feeley noted: "The long-term trend is up and durable... while any mix towards genome over exome in the outpatient setting may introduce some short-term ARR variability, it's ultimately what is best for both patients and our business" .
Risks and Concerns
Near-term headwinds:
- Investment cycle: Q1 2026 will be "close to breakeven" as OpEx ramps ahead of revenue
- Extended adoption timeline: General pediatrics meaningful contribution 18-24 months away
- Reimbursement variability: New call points may initially show 70% denial rates until payer history builds
- Seasonality: Q1 is always the weakest quarter due to deductible resets and weather
Long-term considerations:
- Competitive pressure from other labs expanding into rare disease (though management confident in Infinity moat)
- Regulatory uncertainty around FDA pathway
- Payer pushback on broader genetic testing adoption
Forward Catalysts
Notable: CEO Named to TIME100 Health List
CEO Katherine Stueland was named to the 2026 TIME100 Health list, alongside pioneers like Dr. Monkol Lek and Dr. Rebecca Ahrens-Nicklas from CHOP (who saved Baby KJ with a world-first patient-tailored CRISPR therapy) . CFO Kevin Feeley noted this "signals something important: rare disease and genomic medicine are having their moment. We're decades behind oncology, but we're coming fast" .